LIQUEFACTION OF GASES
LIQUEFACTION OF GASES:When the molecules of a gas are brought closer to each other due to increase in the attractive forces a stage may be reached when the gas changes its phase to liquid. This phenomenon is called Liquefaction of gases. Obviously, an ideal gas cannot be liquefied as there are no interactions in that case. For the liquefaction of Real gases, we can do either of the following:
1. Increase
the pressure on the gas
2. Decrease
the Temperature of the gas
Both these processes will tend to bring the molecules closer and
liquefaction may take place. But it is observed practically that Temperature is the
dominant factor for liquefaction and pressure assumes a secondary role. For
every real gas a temperature is observed above which it cannot be liquefied
even on the application of very high pressures. This characteristic temperature
of a real gas above which it cannot be liquefied is called “Critical
Temperature” represented by TC. To understand this concept in depth, observe the
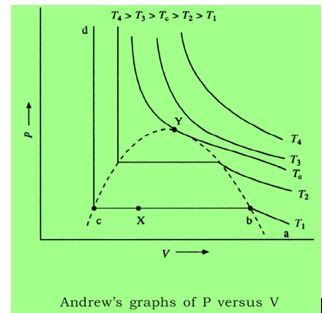
isotherms drawn in the following figure for a real gas at varying temperatures.
LIQUEFACTION OF GASES
We observe the following:
1. At
high Temperatures like T4, the isotherms look just like those of ideal gas.
2. At
low temperatures the curve is entirely different. For example, curve abcd at
temperature T1. As the pressure increases the volume of the gas decreases from
point a to b. Then there is a sudden reduction in volume without much increase
in pressure of the gas from b to c. In this portion liquefaction is taking place
at constant pressure and due to the difference in the properties of gas and
liquid the volume is reducing so much. And after c even on increasing pressure
there is no appreciable decrease in volume as now only liquid is present. So,
ab represents gaseous phase, bc represents liquid-gas coexistence and cd
represents liquid phase.
3. On
carrying out the same process at a higher temperature we observe that the width
of the horizontal portion decreases and the pressure at which liquefaction
takes place also becomes higher. The horizontal portion decreases because at
higher temperatures the properties of liquid approach that of a gas and ΔV = VG
– VL reduces.
4. At
temperature TC the horizontal portion reduces to a mere point at
which liquefaction takes place. Point Y is called critical point ; at this point the boundary between liquid and gas phase (meniscus) disappears indicating that both the phases have identical characteristics.
5. Above
this temperature there is no liquefaction of gases and gas behaves almost ideally.
At critical point Y the tangent is horizontal , so that , dP/dV at critical point will be zero.Thus , the point Y is also called the point of deflection.
At critical point Y the tangent is horizontal , so that , dP/dV at critical point will be zero.Thus , the point Y is also called the point of deflection.
CRITICAL CONSTANTS
Critical
temperature,
TC is the maximum temperature at which a gas can be liquefied that
is a temperature above which liquid cannot exist.
Critical
Pressure, PC
is the minimum pressure required for liquefaction to take place at the critical
condition.
Critical
volume, VC
is the volume occupied by one mole of the gas at critical temperature and
critical pressure.
These parameters are
given by the following expressions:
TC
= 8a/27Rb
PC
= a/27b2
VC
= 3b
Discovery of critical phenomenon by Andrew in 1869 showed that gases can not be liquefied by the application of pressure alone; they must be cooled below their critical temperatures and then subjected to adequate pressure to cause liquefaction.
Principles involved in Liquefaction are :
1. a gas must be at or below its critical temperature. Lower the temperature below the critical value , easier would be the liquefaction.
2. The gas is cooled either by doing external work or by expanding against the internal forces of molecular attraction.
Lower temperature for liquefaction of gases can be achieved by the following techniques :
(a) Cooling by rapid evaporation of a volatileliquid.
(b) Cooling by joule-thomson effect.
(c) Cooling by the adiabatic expansion involving mechanical work
Principles involved in Liquefaction are :
1. a gas must be at or below its critical temperature. Lower the temperature below the critical value , easier would be the liquefaction.
2. The gas is cooled either by doing external work or by expanding against the internal forces of molecular attraction.
Lower temperature for liquefaction of gases can be achieved by the following techniques :
(a) Cooling by rapid evaporation of a volatileliquid.
(b) Cooling by joule-thomson effect.
(c) Cooling by the adiabatic expansion involving mechanical work
Applications of Liquifaction of gases:
- Natural Gas Processing: The liquefaction of natural gas is essential for its storage and transportation. Natural gas is compressed and cooled until it reaches its critical temperature, causing it to become a liquid state, making it easier to transport by tanker ships or pipelines.
- Industrial Refrigeration: The liquefaction of gases such as ammonia, Freon, and carbon dioxide is used as a refrigerant in various industrial processes and refrigeration systems.
- Cooling and Freezing Food: The liquefaction of gases like carbon dioxide and nitrogen is used for quick cooling and freezing of food products in the food processing industry.
- Oxygen Production: The liquefaction of air is used to produce oxygen for medical, industrial and scientific purposes.
- Cryogenics: The liquefaction of gases such as helium and nitrogen is used in cryogenic applications, including superconductivity research, cryogenic fuel storage and rocket propulsion systems.
- Fire Extinguishing: The liquefaction of gases like nitrogen and carbon dioxide is used as a fire extinguishing agent in industries such as chemical plants and oil refineries.
- Welding and Metalworking: The liquefaction of gases like argon and helium is used as a protective shielding gas in various welding and metalworking processes.
- Car Fuel: The liquefaction of gases such as propane and methane is used as a fuel for cars, especially in rural areas where gasoline is not easily available.