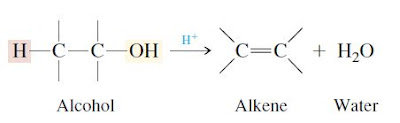
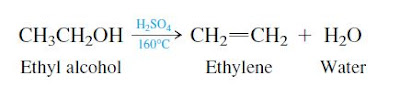In the dehydration of alcohols, the H and OH are lost from adjacent carbons. An acid catalyst is necessary
Before dehydrogenation of ethane became the dominant method, ethylene was prepared by heating ethyl alcohol with sulfuric acid.
Other alcohols behave similarly. Secondary alcohols undergo elimination at lower temperatures than primary alcohols,
Before dehydrogenation of ethane became the dominant method, ethylene was prepared by heating ethyl alcohol with sulfuric acid.
Other alcohols behave similarly. Secondary alcohols undergo elimination at lower temperatures than primary alcohols,