What is Nef reaction
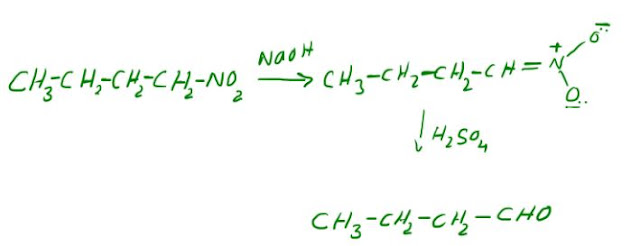
The Nef reaction involves the formation of aldehydes and ketones from primary and secondary nitroparaffins, respectively. Nitroalkanes are first converted to their sodium salts and then hydrolyzed with sulfuric acid to give aldehydes or ketones and nitrous oxide. An example is the formation of butanal from nitrobutane. Sodium hydroxide converts nitrobutane into nitroenolate, which reacts with concentrated sulfuric acid to produce butanal.
Nef Reaction Mechanism
The base removes the α-hydrogen from nitro-alkanes which is acidic because of strong -I effect of the nitro-group and its ability to accommodate negative charge after the removal of proton. The mechanism of Nef reaction is believed to be as follows.