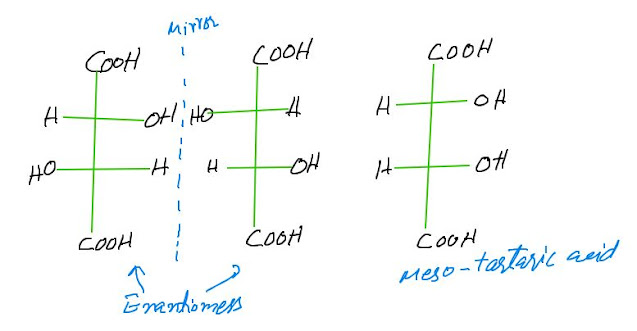
DIASTEREOMERS
Diastereomers are stereoisomers
that are not Mirror image of each other i-e
diastereomers are stereoisomers that are not enantiomers. Consider the steroid isomers of
tartaric acid, meso-tartaric acid is a diastereomer of each of the
enantiomers.
Diastereomers are non-
enantiomeric stereoisomers which have two or more Stereocenters and differ in
the projection of at least one of them.
Glucose,
galactose and mannose are all diastereomers
Diastereomers have a major advantage over the enantiomers from a practical point of view. Diastereomers have different physical properties like m.p. , b.p., solubility, retention time and Rf values and have different rates of reactions even in achiral environments. Standard techniques like crystallization, distillation or chromatography can therefore be used to separate diastereomeric mixtures.
Diastereomers--- Both molecules must have the same molecular formula and Both molecules must have the same atom connectivity. However they have different 3D shape and are non-superimpossible in space.
It is important to note that in general a molecule can have only one enantiomer, but it may have many diastereomers.
When two diastereomers differ in the stereochemistry at only one stereocenter then these are called Epimers.
The diastereomers which differ in the configuration at C1 (first carbon in a molecule) are called anomers.